Okay, let’s talk lithium batteries — not the hype machine type, not the “BYD's go vroom” level of battery discourse. I mean the deep, materials-science-tier grind that actually shapes the future of high-density energy.
Because here’s the thing: the tech we’re relying on to scale EVs, phones, and massive grid batteries? It still doesn't work at scale. LiFePO4 and NCM cathodes hit a wall at around 300 Wh/kg. If you want more, you're not getting it with the current cast of characters. Enter: Li-rich Mn-based oxides, or LRMOs.
They’ve got potential, there were some issue and now, someone had the audacity to slap silica-coated Silver nanowires on them — and it works. Let's unpack this.
Why Li-Rich Mn-Based Oxides Aren’t Already Running the World
LRMOs are promising for lithium-ion cathodes because of their beastly theoretical capacities — over 250 mAh/g and cutoff voltages pushing 4.8V. That’s a straight-up win in the battery world!
But the problems hit like a freight train:

The main culprit? Structural instability and oxygen loss during repeated charge/discharge cycles. Think of it like a party house falling apart every time you throw a rave. Great for one night. Terrible after the tenth.
The Fix We Didn’t Know We Needed
The study doesn’t reinvent the wheel. It just wraps it in silica-coated silver nanowires (AgNWs@SiO₂) and somehow makes it roadworthy at 150mph.
Here’s what these nanowires do when added to LRMO:

And no, this isn’t lab fluff. The researchers straight up compared AgNWs@SiO₂-modified LRMO against both the raw LRMO and AgNWs-only versions. Guess who won?
Performance Stats of Silver Nanowires
AgNWs@SiO₂-modified LRMO:

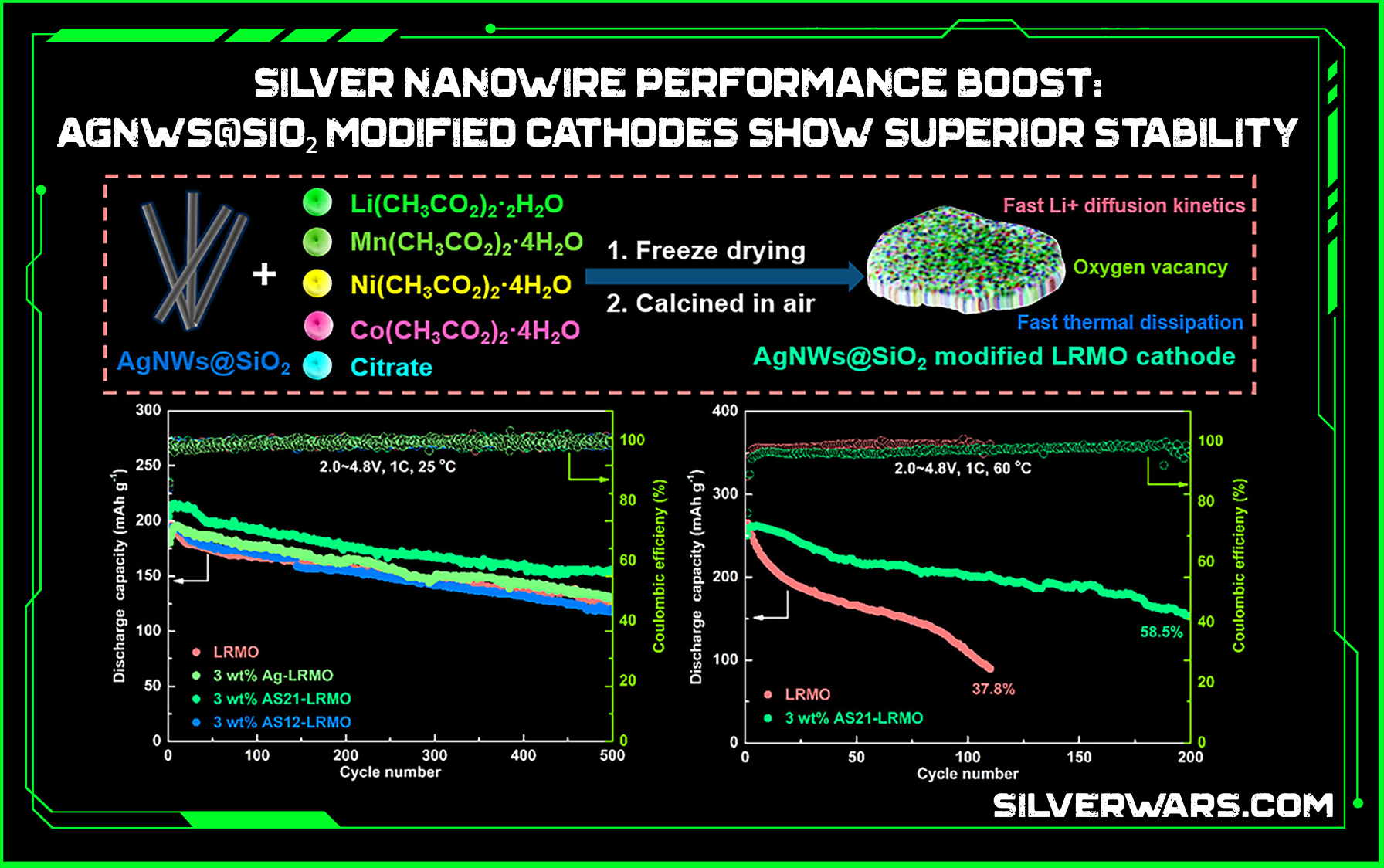
That 50.6% retention at 3C after 1000 cycles? Clip that Chat. Most cathodes would be gasping for life after 400. This one’s still jogging.
Why This Actually Matters
This isn’t about squeezing out a couple more mAh/g. This is about making lithium batteries survive the brutal conditions of high-demand use — high current, high temp, long life. You want a battery that can:
- Handle fast charging without cooking itself
- Last more than 500 charge cycles without turning into a paperweight
- Deliver consistent voltage without dropping off a cliff
And this silver-silica nanowire combo gets us a step closer to that. It’s not a full-blown miracle, but it patches up the biggest pain points in LRMO tech.

Cathode Design, Simplified
Here’s how they actually built this thing:
- Start with LRMO: Li₁.₂Mn₀.₅₄Ni₀.₁₃Co₀.₁₃O₂
- Introduce Silver Nanowires
- Coat them in SiO₂ to prevent oxidation
- Freeze-dry + high-temperature calcination
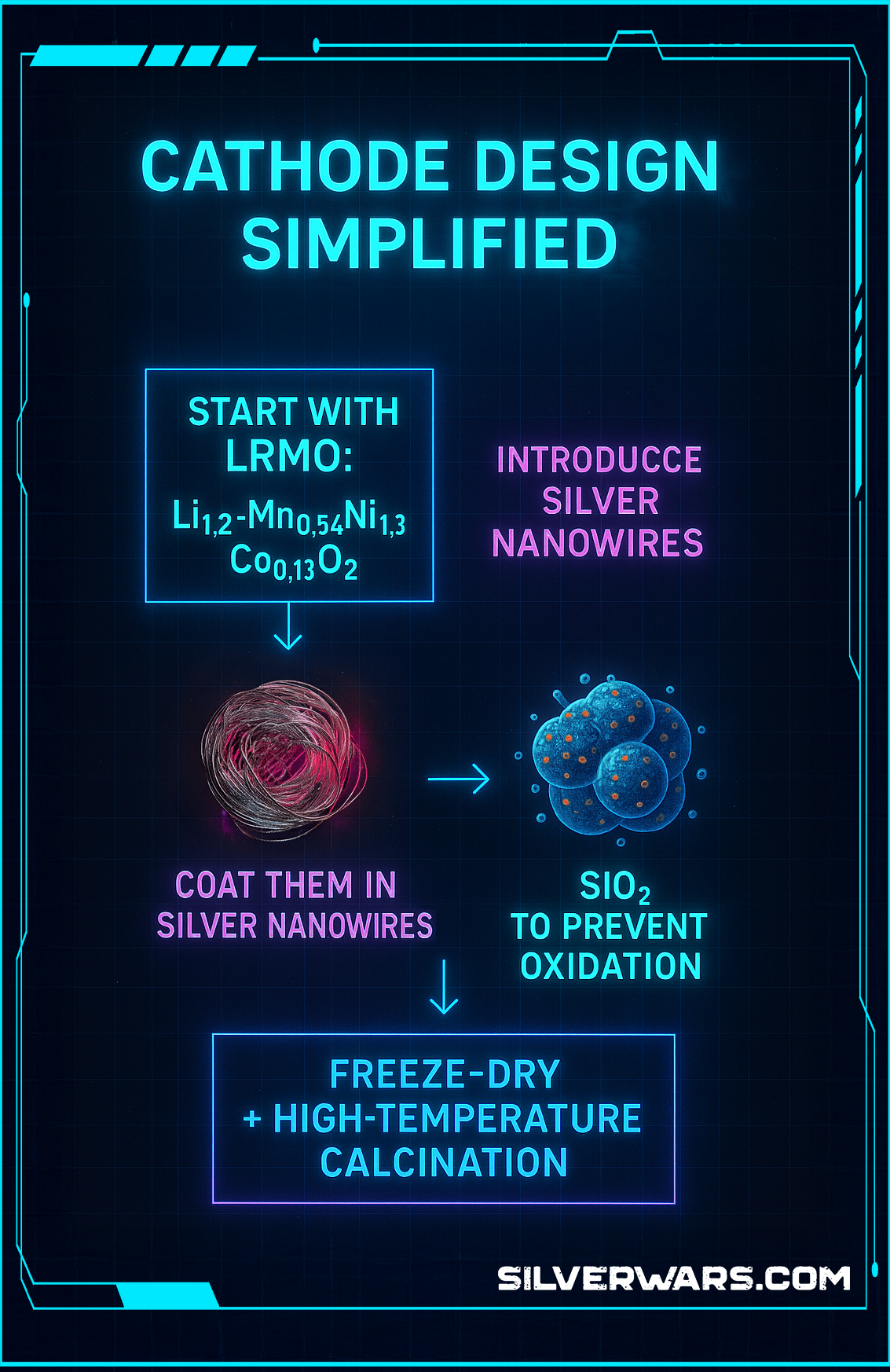
Why the silica? Because silver nanowires alone are reactive and unstable at high heat. The SiO₂ layer keeps them in check, prevents clumping, and locks them in place during the calcination phase.
The result? Uniform dispersion, increased structural stability, and heat control baked into the cathode.

Full-Cell Compatibility
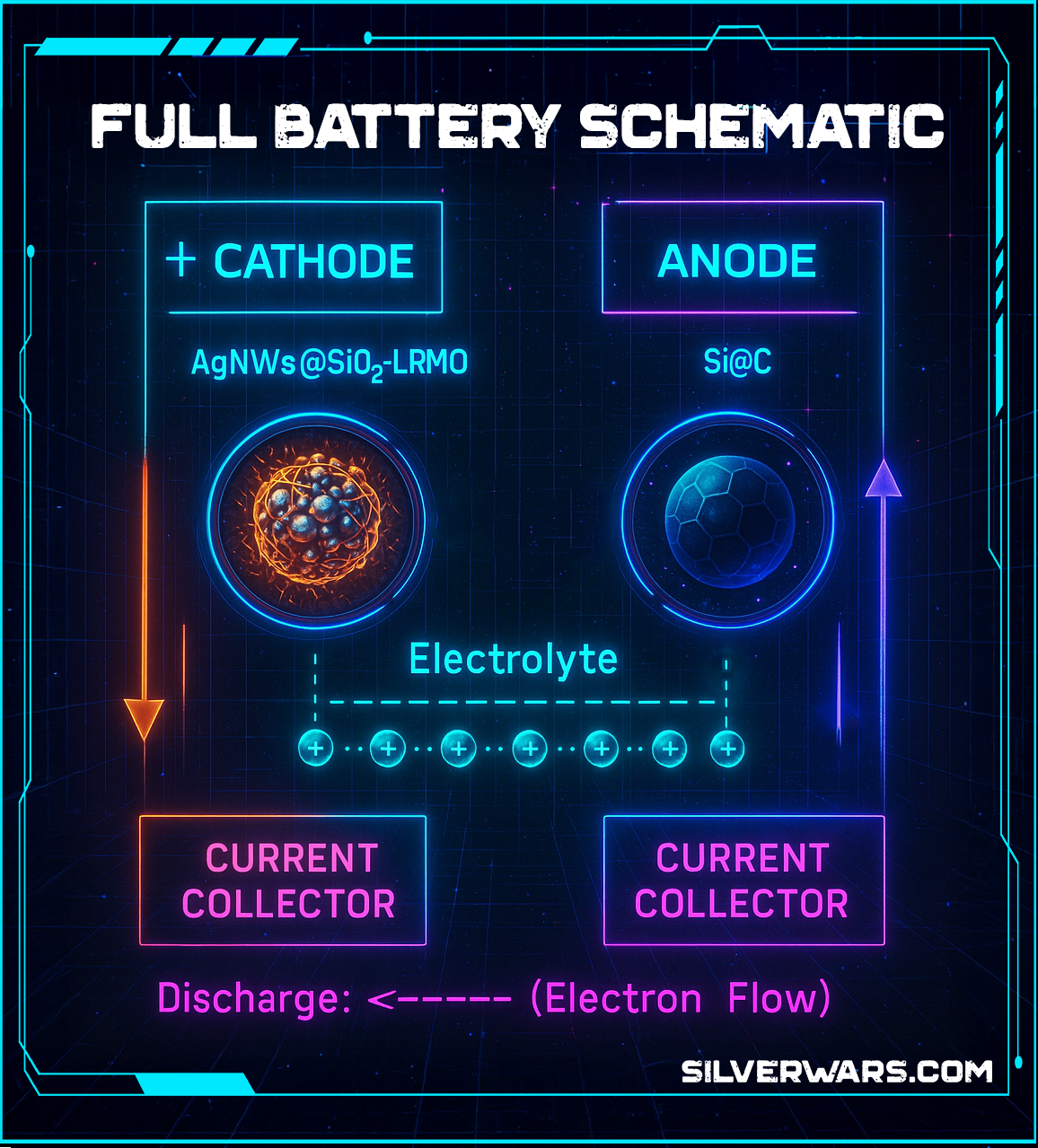
Just in case you were about to ask “but does it actually work in a real battery,” yes. They paired the AgNWs@SiO₂-modified LRMO cathode with a Si@C anode and got solid full-cell performance. That means this isn’t some theoretical academic flex. It's closer to real-world implementation than half the stuff hyped in industry whitepapers.

TL;DR
- LRMOs have insane potential but suffer from structural and voltage instability.
- Silica-coated silver nanowires (AgNWs@SiO₂) massively boost performance:
- Faster ion transport
- Better thermal control
- Less voltage fade
- Longer cycle life
- AgNWs@SiO₂-modified LRMO hit 71.6% retention after 500 cycles at 25°C and outperformed raw LRMO at high temp and high rate.
- Full-cell testing? Done. It works.









